
Regulation of cell growth and survival by growth factors
Targeting the metabolic liabilities of cancer cells by modulating endolysosomal trafficking and nutrient access.
The Edinger lab uses interdisciplinary, collaborative approaches to overcome challenging problems in the field of cancer metabolism. Rather than focusing on individual anabolic enzymes and metabolites, we target intracellular trafficking pathways to more globally disrupt nutrient acquisition. This holistic approach should limit the development of drug resistance and produce new therapies that are broadly active against cancers with many different driver mutations. By starting with endogenous molecules that have been optimized by evolution to balance growth suppression with toxicity, we can design compounds that are safe for normal cells despite their multi-faceted, anti-cancer actions.
Goal
Develop innovative cancer therapies that are less toxic and more effective than currently available drugs both when used as single agents and in combination with existing therapies.
Innovation
We target apical, regulatory nodes to disable multiple oncogenic pathways simultaneously. Using drug-like variants of natural molecules that control growth limits the toxicity of this approach.
Approach
By expanding our understanding of how cancer cells acquire and process nutrients, we hope to uncover new targets for cancer therapies and increase our knowledge of how cells respond to stress.
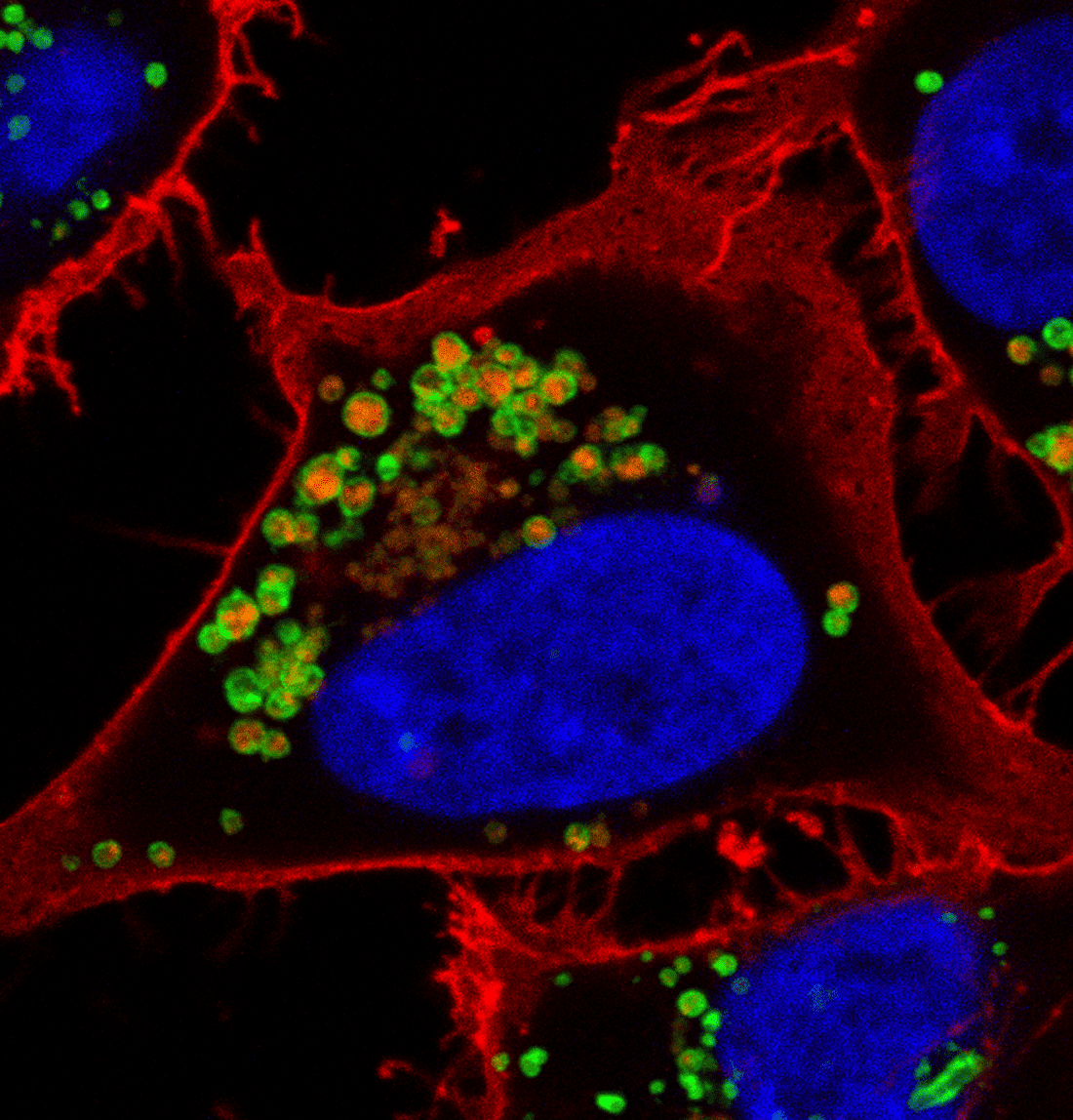
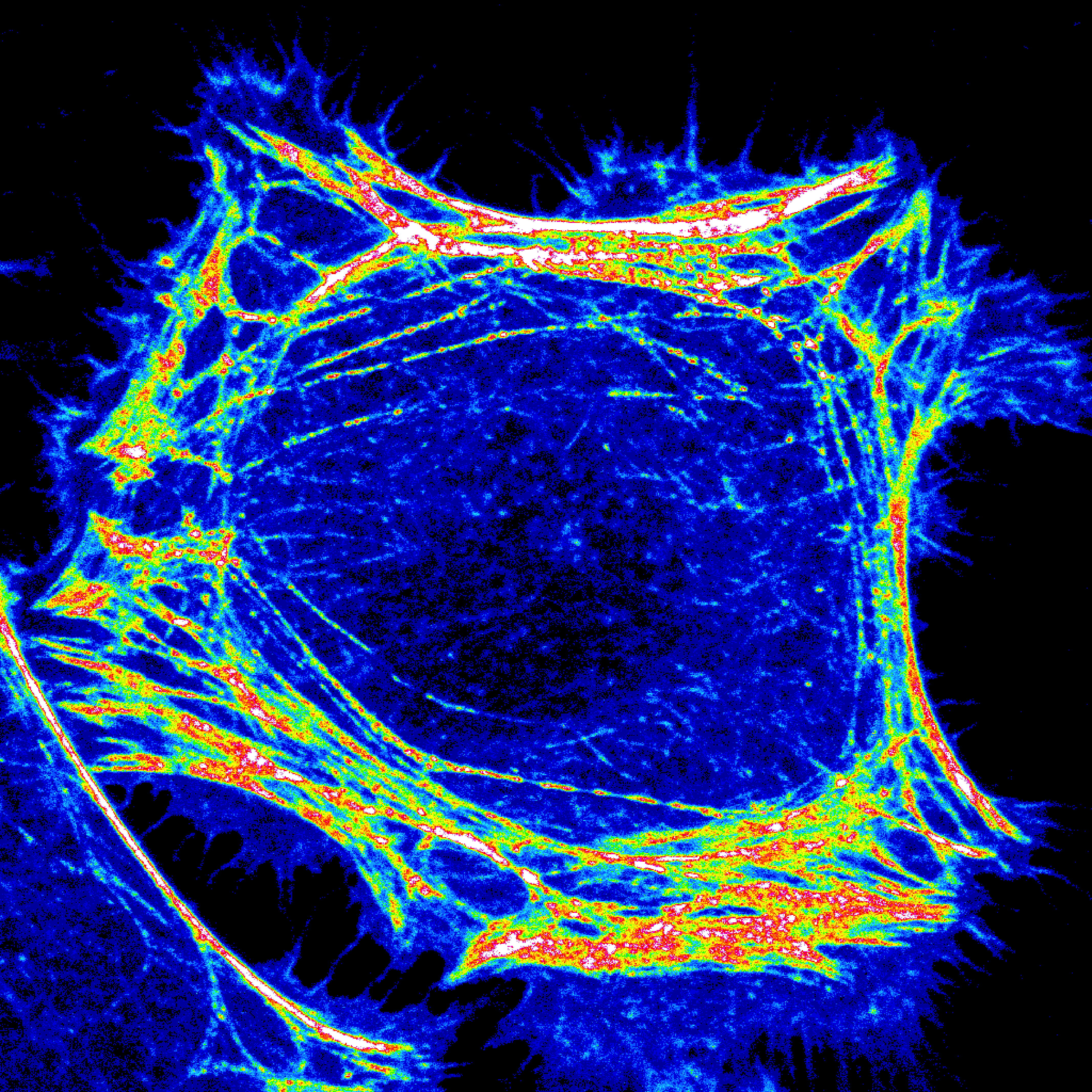
Current research in the Edinger lab focuses on:
Sphingolipids and Growth Control
Sphingolipids have been called “tumor suppressor lipids” because they slow cell growth, induce differentiation, and trigger programmed cell death. Sphingolipids were named after the Sphinx due to their enigmatic actions. We dissect the complex molecular mechanisms underlying the ability of sphingolipids to restrict cancer initiation and growth with a focus on how these lipids control the endocytosis of cell-surface nutrient transporter proteins and the decision point where these proteins are either recycled back to the plasma membrane or sent to the lysosome for degradation. This work has revealed evolutionarily-conserved mechanisms for growth control that are disrupted in cancer cells.
Drug Development and Chemical Biology
In collaboration with chemist Stephen Hanessian, PhD, we have developed novel synthetic sphingolipids with anti-cancer properties. Like natural sphingolipids, our compounds slow cell growth, induce differentiation, and kill cancer cells. Unlike endogenous sphingolipids, our drug-like versions are resistant to metabolism, water-soluble, and orally bioavailable. We have demonstrated that these molecules are safe and effective in models of colon, prostate, and breast cancer. This work has led to the launch of a start-up biotech company that is moving these molecules to clinical trials in cancer patients. Through chemoproteomics conducted in collaboration with Pierre Thibault’s group at the University of Montreal, we are using our panel of analogs to dissect the protein targets responsible for the multifaceted effects of sphingolipids.
Macropinocytosis and Nutrient Scavenging
Nutrient delivery to tumor cells is limited by abnormal, leaky blood vessels. When glucose, amino acids, and other building blocks are limited, cancer cells scavenge macromolecules from the tumor microenvironment and then degrade them into subunits that are used to fuel growth. Macropinocytosis is a non-specific, bulk uptake process through which macromolecules can be engulfed by membrane ruffles that close into large (0.2 – 5 μm) macropinosomes. Our work has shown that prostate and breast cancers rely on macropinocytosis to support their growth and survival. Inhibitors of macropinocytosis limit tumor growth in mice. We are dissecting the molecular signals that control macropinocytosis and exploring its role in tumor growth and progression with the goal of developing safe and effective inhibitors that can be used therapeutically.
Anti-sense Oligonucleotide Potentiation
leaky blood vessels. When glucose, amino acids, and other building blocks are limited, cancer cells scavenge macromolecules from the tumor microenvironment and then degrade them into subunits that are used to fuel growth. Macropinocytosis is a non-specific, bulk uptake process through which macromolecules can be engulfed by membrane ruffles that close into large (0.2 – 5 μm) macropinosomes. Our work has shown that prostate and breast cancers rely on macropinocytosis to support their growth and survival. Inhibitors of macropinocytosis limit tumor growth in mice. We are dissecting the molecular signals that control macropinocytosis and exploring its role in tumor growth and progression with the goal of developing safe and effective inhibitors that can be used therapeutically.
Recent Publications
- Selwan E and AL Edinger (2017). Branched chain amino acid metabolism and cancer: the importance of keeping things in context. Translational Cancer Research, 6(Suppl 3):S578-S584. doi: 10.21037/tcr.2017.05.05 (commentary on Science 353:1161-5 (2016).
- Kallenborn-Gerhardt W, Möser CV, Lorenz JE, Steger M, Heidler J, Scheving R, Petersen J, Kennel L, Flauaus C, Lu R, Edinger AL, Tegeder I, Geisslinger G, Heide H, Wittig I, PhD, and Schmidtko A (2017). Rab7 – a novel redox target that modulates inflammatory pain processing. PAIN, in press. PMID: 28394828
- McCracken AN, McMonigle RJ, Tessier J, Fransson R, Perryman MS, Chen B, Keebaugh A, Selwan E, Barr SA, Kim SM, Roy SG, Liu G, Fallegger D, Sernissi L, Brandt C, Moitessier N, Snider AJ, Clare S, Müschen M, Huwiler A, Kleinman MT, Hanessian S, and AL Edinger (2017). Phosphorylation of a constrained azacyclic FTY720 analog enhances anti-leukemic activity without inducing S1P receptor activation. Leukemia. 31(3):669-677. PMID: 27573555
- Jaber N, Mohd-Naim N, Wang Z, DeLeon JL, Kim SM, Zhong H, Sheshadri N, Dou Z, Edinger AL, Du G, Braga VMM, and W-X Zong (2016). Vps34 regulates Rab7 and late endocytic trafficking through recruitment of the GTPase activating protein Armus. Journal of Cell Science, 129(23):4424-4435.
- Kim SM, Roy SG, Chen B, Nguyen T, McMonigle RJ, McCracken AN, Finicle BT, Zhang Y, Kofuji S, Hou J, Selwan E, Nguyen T, Ravi A, Ramirez MU, Wiher T, Guenther GG, Kono M, Sasaki AT, Weisman LS, Potma EO, Tromberg BJ, Hanessian S, and AL Edinger (2016). Targeting cancer metabolism by simultaneously disrupting parallel nutrient access pathways. Journal of Clinical Investigation 126(11):4088-4108. PMID: 27669461
- Perryman M, Tessier J, Wiher T, O’Donoghue H, McCracken AN, Kim SM, Nguyen GD, Simitan GS, Viana M, Rafelski S, Edinger AL# and S Hanessian# (2016). Effects of stereochemistry, saturation, and hydrocarbon chain length on the ability of constrained azacyclic sphingolipids to trigger nutrient transporter down-regulation, vacuolation, and cancer cell death. (# = co-corresponding authors) Bioorganic and Medicinal Chemistry 24:4390-7. PMID: 27475534
- Walker WP, Oehlerb A, Edinger AL, Wagner K-U, and TM Gunn (2016). Oligodendroglial deletion of ESCRT-I component TSG101 causes spongiform encephalopathy. Biology of the Cell, 108(11): 324-337. PMID: 27406702.
- Safaiyan S, Snaidero N, Edinger AL, Jung S, M Simons (2016). Age-related myelin fragmentation overwhelms the clearance function of microglia during normal aging. Nature Neuroscience 19:995-8. PMID: 27294511
- Klionsky D . . . Edinger AL . . . et al. (2016). Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 12(1):1-222. PMC4835977
- Selwan EM, Finicle BT, Kim SM, and AL Edinger (2016). Attacking the supply wagons to starve cancer cells to death. FEBS Lett. 590:885-907. PMID: 26938658
- Chen B*, Roy SG*, McMonigle RJ, Keebaugh A, McCracken AN, Selwan E, Fransson R, Fallegger D, Huwiler A, Kleinman MT, Edinger AL# and S Hanessian# (2016). Azacyclic FTY720 Analogues that Limit Nutrient Transporter Expression but Lack S1P Receptor Activity and Negative Chronotropic Effects Offer a Novel and Effective Strategy to Kill Cancer Cells in vivo. ACS Chemical Biology,11(2):409-14. (* = co-first authors, # = co-corresponding authors)
- McCracken AN and AL Edinger (2015). Targeting cancer metabolism at the plasma membrane by limiting amino acid access through SLC6A14. Biochem. J., 470(3):e17-9.
- Holden JK, Kang S, Beasley FC, Cinelli MA, Li h, Roy SG, Dejam D, Edinger AL, Nizet V, Silverman RB, and TL Poulos (2015). Nitric Oxide Synthase as a Target for Methicillin Resistant Staphylococcus aureus. Chemistry & Biology 22:785-92. PMID: 26091171
- Pone EJ, Lam T, Edinger AL, Xu Z, and P Casali (2015). B Cell Rab7 Mediates Induction of AID Expression and Class-switching in T-dependent and T-independent Antibody Responses. Journal of Immunology 194:3065-78. PMID: 25740947
- Guenther GG*, Liu G*, Ramirez MU, McMonigle RJ, McCracken AN, Kim SM, Joo Y, Ushach I, Nguyen NL, and AL Edinger (2014). Loss of TSC2 confers resistance to ceramide and nutrient deprivation. Oncogene 33:1776-1787. PMID: 23604129
- Fransson R*, McCracken AN*, Chen B, McMonigle RJ, Edinger AL# and S Hanessian#. Design, Synthesis, and Anti-leukemic Activity of Stereochemically Defined Constrained Analogs of FTY720 (Gilenya). ACS Medicinal Chemistry Letters, in press. (* = co-first authors, # = co-corresponding authors)
- Guenther GG*, Liu G*, Ramirez MU, McMonigle RJ, McCracken AN, Kim SM, Joo Y, Ushach I, Nguyen NL, and AL Edinger (2013). Loss of TSC2 confers resistance to ceramide and nutrient deprivation. Oncogene (ePub ahead of print).
- Roy SG, Stevens MW, So L and AL Edinger (2013). Reciprocal effects of Rab7 deletion in activated and neglected T cells. Autophagy vol. 9, issue 7 (ePub ahead of print).
- McCracken AN and AL Edinger (2013). Nutrient transporters: the Achilles’ heel of anabolism. Trends in Endocrinology and Metabolism 24:200-8.
- Klionsky DJ . . . Edinger AL . . . (and others) (2012). Guidelines for the use and interpretation of assays for monitoring autophagy in higher eukaryotes. Autophagy 8:445-544.
- Chen J, Narayan S, Edinger AL, and MJ Bennett (2012). Flow injection tandem mass spectrometric measurement of ceramides of multiple chain lengths in biological samples. J Chromatogr B Analyt Technol Biomed Life Sci. 883-884: 136-140.
- Romero Rosales K, Singh G, Wu K, Chen J, Lilly MB, Peralta ER, Janes MR, Siskind LJ, Bennett MJ, Fruman DA, and AL Edinger (2011). Sphingolipid-based drugs selectively kill cancer cells by down-regulating nutrient transporter proteins. Biochem. J. 439:299-311.
- AL Edinger (2011). Unequal in the absence of death: A novel screen identifies cytotoxic compounds selective for cells with activated Akt. Cancer Biol Ther. 10:1262-5.
- Walsh CM and AL Edinger (2010). The complex interplay between autophagy, apoptosis, and necrotic signals promotes T-cell homeostasis. Immunol. Rev. 236:95-109.
- Peralta EP, Martin BC, and AL Edinger (2010). TBC1D15 is a selective Rab7 GTPase activating protein but mammalian Vps39 does not modulate Rab7 GTP binding status. J. Biol. Chem. 285:16814-21.http://www.ncbi.nlm.nih.gov/pubmed/20363736 This paper identifies proteins that regulate lysosomal dynamics and shows that they modulate growth factor dependence.
- Romero KR, Peralta EP, Guenther GG, Wong SY, and AL Edinger (2009). Rab7 activation by growth factor withdrawal contributes to the induction of apoptosis. Molec. Biol. of the Cell 20:2831-40.http://www.ncbi.nlm.nih.gov/pubmed/19386765?itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_RVDocSum&ordinalpos=1 This paper demonstrates that Rab7 activity is regulated by growth factors and that activating Rab7 can kill cells.
- AL Edinger (2008). Starvation in the midst of plenty: making sense of ceramide-induced autophagy by analyzing nutrient transporter expression. Biochem Soc Trans. 2009 Feb;37(Pt 1):253-8. http://www.ncbi.nlm.nih.gov/pubmed/19143642?itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_RVDocSum&ordinalpos=4This review helps to place our recent studies of the effect of ceramide on nutrient transporter expression in context.
- Guenther GG, Peralta EP, Romero KR, Wong SY, Siskind, LJ and AL Edinger (2008). Ceramide starves cells to death by down-regulating nutrient transporter proteins. Proc. Natl. Acad. Sci. 105, 17402–17407. http://www.ncbi.nlm.nih.gov/pubmed/18981422?ordinalpos=1itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_DefaultReportPanel.Pubmed_RVDocSumThis paper establishes for the first time that ceramide kills cells through a bioenergetic mechanism.
- Edinger, AL (2007). Controlling cell growth and survival through regulated nutrient transporter expression. Biochem. J. 406:1-12.http://www.ncbi.nlm.nih.gov/pubmed/17645414?ordinalpos=3&itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_DefaultReportPanel.Pubmed_RVDocSumComprehensive review of regulated nutrient transporter expression and its role in cellular growth control.
